FDA issues urgent recall of mislabeled pain medication over life-threatening risks
- Replies 0
When it comes to everyday health routines, most people hardly pause to double-check the details. Pill bottles sit in cabinets, trusted without question, each label assumed to mean what it says.
But sometimes, even the containers we’ve grown used to seeing and handling without a second thought can hide risks that most people would never anticipate.
That sense of reliability and safety has now been unsettled by a fresh alert issued by federal regulators, leaving many to wonder what might come next.
The Food and Drug Administration has announced an urgent recall after a mislabeled prescription drug raised alarms over safety risks.
Unichem Pharmaceuticals, based in New Jersey, confirmed that one lot of Cyclobenzaprine Hydrochloride Tablets USP in 10 mg doses was incorrectly labeled.
Instead of the intended muscle relaxant, some bottles actually contained Meloxicam 7.5 mg tablets, a nonsteroidal anti-inflammatory drug.
This mix-up means patients could unknowingly take the wrong medication, a situation federal officials say carries a “reasonable probability” of life-threatening reactions.
Cyclobenzaprine, often sold under the brand names Amrix or Fexmid, is typically prescribed for short-term relief of muscle spasms.
By contrast, Meloxicam—sold as Mobic or Vivlodex—is used to treat arthritis-related pain and inflammation, often in patients with long-term conditions.
The FDA warned that people taking Meloxicam in error could face serious complications, ranging from cardiovascular and gastrointestinal damage to kidney injury, anaphylaxis, and severe skin reactions.
Those who already take other NSAIDs or live with certain comorbidities are believed to face the highest risk.
Also read: Traveling with medication? Some over-the-counter drugs could cause trouble abroad
So far, there have been no adverse reactions officially reported in connection with this recall, but the potential danger has prompted swift action.
The bottles in question are tied to lot number GMML24026A, marked with an expiration date of September 2027 and labeled with NDC 29300-415-19.
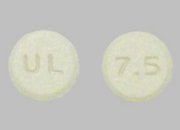
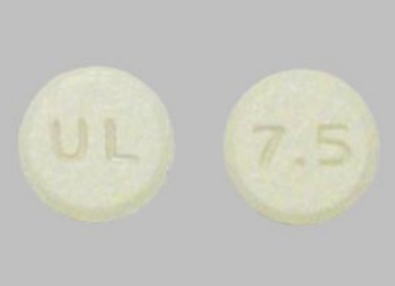
Cyclobenzaprine tablets are normally round, blue, and film-coated with “U” and “12” engraved on their sides. By comparison, Meloxicam tablets are light yellow, engraved with “U & L” on one side and “7.5” on the other, making visual checks an essential safeguard for patients.
Distribution of the mislabeled medication extended nationwide, reaching both pharmacies and consumers. Regulators have instructed that all bottles from this lot be immediately pulled from circulation and returned through Inmar, the recall management company.
Also read: Health warning issued over popular bedroom item linked to toxic exposure
Retail pharmacies are directed to stop dispensing from this batch and to notify any customers who may have already received it. Patients in possession of the recalled product are urged to return it to their point of purchase rather than attempting to use it.
The recall serves as a sharp reminder of how heavily millions rely on accuracy in pharmaceutical labeling. In 2024 alone, more than 24 million prescriptions were written for Cyclobenzaprine, while Meloxicam was prescribed over 20 million times to nearly 7 million Americans.
Both drugs play an important role in pain management, but substituting one for the other could lead to devastating outcomes. Regulators stressed that while medication errors are rare, vigilance by both providers and patients remains critical to ensuring safety.
Read next: Urgent recall affects 2 popular heart medications sold nationwide

If you discovered the pills in your medicine cabinet looked different than usual, would you trust the label—or stop and check before taking them? Share your perspective on whether enough safeguards exist to prevent dangerous mix-ups like this one.
But sometimes, even the containers we’ve grown used to seeing and handling without a second thought can hide risks that most people would never anticipate.
That sense of reliability and safety has now been unsettled by a fresh alert issued by federal regulators, leaving many to wonder what might come next.
The Food and Drug Administration has announced an urgent recall after a mislabeled prescription drug raised alarms over safety risks.
Unichem Pharmaceuticals, based in New Jersey, confirmed that one lot of Cyclobenzaprine Hydrochloride Tablets USP in 10 mg doses was incorrectly labeled.
Instead of the intended muscle relaxant, some bottles actually contained Meloxicam 7.5 mg tablets, a nonsteroidal anti-inflammatory drug.
This mix-up means patients could unknowingly take the wrong medication, a situation federal officials say carries a “reasonable probability” of life-threatening reactions.

FDA issues urgent recall of mislabeled pain medication over life-threatening risks. Image source: walgreens
Cyclobenzaprine, often sold under the brand names Amrix or Fexmid, is typically prescribed for short-term relief of muscle spasms.
By contrast, Meloxicam—sold as Mobic or Vivlodex—is used to treat arthritis-related pain and inflammation, often in patients with long-term conditions.
The FDA warned that people taking Meloxicam in error could face serious complications, ranging from cardiovascular and gastrointestinal damage to kidney injury, anaphylaxis, and severe skin reactions.
Those who already take other NSAIDs or live with certain comorbidities are believed to face the highest risk.
Also read: Traveling with medication? Some over-the-counter drugs could cause trouble abroad
So far, there have been no adverse reactions officially reported in connection with this recall, but the potential danger has prompted swift action.
The bottles in question are tied to lot number GMML24026A, marked with an expiration date of September 2027 and labeled with NDC 29300-415-19.
Cyclobenzaprine tablets are normally round, blue, and film-coated with “U” and “12” engraved on their sides. By comparison, Meloxicam tablets are light yellow, engraved with “U & L” on one side and “7.5” on the other, making visual checks an essential safeguard for patients.
Distribution of the mislabeled medication extended nationwide, reaching both pharmacies and consumers. Regulators have instructed that all bottles from this lot be immediately pulled from circulation and returned through Inmar, the recall management company.
Also read: Health warning issued over popular bedroom item linked to toxic exposure
Retail pharmacies are directed to stop dispensing from this batch and to notify any customers who may have already received it. Patients in possession of the recalled product are urged to return it to their point of purchase rather than attempting to use it.
The recall serves as a sharp reminder of how heavily millions rely on accuracy in pharmaceutical labeling. In 2024 alone, more than 24 million prescriptions were written for Cyclobenzaprine, while Meloxicam was prescribed over 20 million times to nearly 7 million Americans.
Both drugs play an important role in pain management, but substituting one for the other could lead to devastating outcomes. Regulators stressed that while medication errors are rare, vigilance by both providers and patients remains critical to ensuring safety.
Read next: Urgent recall affects 2 popular heart medications sold nationwide
Key Takeaways
- The FDA recall involves bottles labeled as Cyclobenzaprine 10 mg that actually contain Meloxicam 7.5 mg tablets.
- Officials warned of a “reasonable probability” of life-threatening reactions, including cardiovascular, renal, gastrointestinal, and allergic complications.
- The affected lot is GMML24026A with expiration September 2027, distributed nationwide through pharmacies and retailers.
- Consumers and pharmacies are advised to return mislabeled products through Inmar and avoid dispensing or consuming the recalled tablets.